(a)(i) Give two uses of chlorine.
(ii) State the action of chlorine on moist blue litmus paper
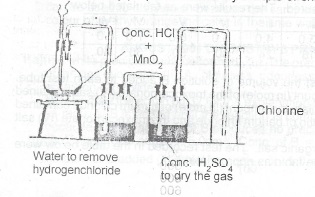
(b) Draw a labelled diagram for the laboratory preparation of a dry sample of chlorine
(c) State the type of reaction involved between chlorine and (i) aqueous iron (II) chloride;
(ii) propane. Write an equation for each reaction and name the product formed in (c)(ii).
(d) Consider the reactions the following equations: Cl\(_{2(g)}\) + 2Br\(^-_{(aq)}\) \(\to\) 2Cl\(^-_{(aq)}\) + Br\(_{2(g)}\)
F\(_{2(g)}\) + 2Cl\(^-_{(aq)}\) ---> 2F\(^-_{(aq)}\) + Cl\(_{2(g)}\)
From the equations, arrange bromine, chlorine and fluorine in increasing order of oxidizing ability. Give the reason for your answer.
(ii) State the action of chlorine on moist blue litmus paper
(b) Draw a labelled diagram for the laboratory preparation of a dry sample of chlorine
(c) State the type of reaction involved between chlorine and (i) aqueous iron (II) chloride;
(ii) propane. Write an equation for each reaction and name the product formed in (c)(ii).
(d) Consider the reactions the following equations: Cl\(_{2(g)}\) + 2Br\(^-_{(aq)}\) \(\to\) 2Cl\(^-_{(aq)}\) + Br\(_{2(g)}\)
F\(_{2(g)}\) + 2Cl\(^-_{(aq)}\) ---> 2F\(^-_{(aq)}\) + Cl\(_{2(g)}\)
From the equations, arrange bromine, chlorine and fluorine in increasing order of oxidizing ability. Give the reason for your answer.
Take Free Practice Test On 2026 JAMB UTME, Post UTME, WAEC SSCE, GCE, NECO SSCE
Correct Answer: Option n
Explanation:

(a) Chlorine is used as a bleaching and in the manufacture of bleaching agents. Chlorine is also used as domestic antiseptic solution such as sodium oxochlorate
(i) NaOCI.
(ii) Chlorine is an acidic gas and therefore, in its contact with moist blue litmus paper will first turn the paper red before bleaching it.
(b) Laboratory preparation of a dry sample of chlorine \(MnO_2{(s)} + 4HCl_{(aq)} \to MnCl_{2(aq)} + 2H_2O_{(l)} + Cl_{2(g)}\)
(c)(i) \(2FeCl_{2(g)} + Cl_{2(g)} \to 2FeCl_{3(aq)}\) (Redox reaction)
(green) (red brown)
(ii) \(H_2C=Ch-CH_{3(g)} + Cl_{_(g)}\) \(\to\) \(CH_2ClCHClCH_3\) (addition type reaction)
(ii) The product in (c)(ii) is called 1, 2-dichloropropane.
(d)
Cl\(_{2(g)}\) oxides 2Br\(^-\) to Br\(_{2(l)}\) while F\(_{2(g)}\) oxides 2Cl\(^{-}\) to Cl\(_{2(g)}\)
(a) Chlorine is used as a bleaching and in the manufacture of bleaching agents. Chlorine is also used as domestic antiseptic solution such as sodium oxochlorate
(i) NaOCI.
(ii) Chlorine is an acidic gas and therefore, in its contact with moist blue litmus paper will first turn the paper red before bleaching it.
(b) Laboratory preparation of a dry sample of chlorine \(MnO_2{(s)} + 4HCl_{(aq)} \to MnCl_{2(aq)} + 2H_2O_{(l)} + Cl_{2(g)}\)
(c)(i) \(2FeCl_{2(g)} + Cl_{2(g)} \to 2FeCl_{3(aq)}\) (Redox reaction)
(green) (red brown)
(ii) \(H_2C=Ch-CH_{3(g)} + Cl_{_(g)}\) \(\to\) \(CH_2ClCHClCH_3\) (addition type reaction)
(ii) The product in (c)(ii) is called 1, 2-dichloropropane.
(d)
Cl\(_{2(g)}\) oxides 2Br\(^-\) to Br\(_{2(l)}\) while F\(_{2(g)}\) oxides 2Cl\(^{-}\) to Cl\(_{2(g)}\)