(a)(i) Define the term polymerization.
(ii) List the three conditions required for the polymerization on of ethene.
(iii) State the property which is common to compounds that can be easily polymerized
(b) Write appropriate equations to show how the following can be obtained from propan-1-ol in the labouratory
(i) propene;
(ii) propylmethanoate. State the type of reaction involved in each case.
(c)(i) A compound contains 40.0% carbon, 6.7% hydrogen and 53.3% oxygen. Determine its molecular formula if its molar mass is 180 (H = 1, C = 12, O = 16)
(ii) Explain why ethanoic acid boils at a much higher temperature than butane even though their molar masses almost equal.
(ii) List the three conditions required for the polymerization on of ethene.
(iii) State the property which is common to compounds that can be easily polymerized
(b) Write appropriate equations to show how the following can be obtained from propan-1-ol in the labouratory
(i) propene;
(ii) propylmethanoate. State the type of reaction involved in each case.
(c)(i) A compound contains 40.0% carbon, 6.7% hydrogen and 53.3% oxygen. Determine its molecular formula if its molar mass is 180 (H = 1, C = 12, O = 16)
(ii) Explain why ethanoic acid boils at a much higher temperature than butane even though their molar masses almost equal.
Take Free Practice Test On 2026 JAMB UTME, Post UTME, WAEC SSCE, GCE, NECO SSCE
Correct Answer: Option n
Explanation:
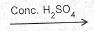
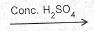
(a) (i) Polymerization is the combination of many molecules of the same kind to form a complex molecule Conc. H\(_2\)SO\(_4\) without gain or loss of material.
(b)(i) \(CH_3CH_2CH_2OH_{(aq)}\) \(CH_2CHCH_3\) + (\(H_2SO_4 + H_2O\)).
(ii) \(CH_3CH_2CH_2OH_{(aq)}\) + CHOOH --> \(CH_3CH_2CH_2OH_{(l)}\) + H\(_2\)O\(_{(l)}\)
(i) & (ii) are respectively dehydration and condensation type of reactions.
(c)(i) Carbon 40.O% = \(\frac{40}{12}\) = 3.33 moles ; Hydrogen 6.7% moles
\(\frac{6.7}{1} = 6.7\) moles
Oxygen 53.3% = \(\frac{53.3}{16}\) = 3.33 moles
Divide through by smaller number
i.e. C \(\frac{33}{3.33}\) : H \(\frac{6.7}{3.33}\) : O \(\frac{3.33}{3.33}\)
C = 1: H = 2.0: O = 1
Empirical formula = CH\(_2\)O(CH\(2\)O)x = 180
i.e. (30)x = 180
x = \(\frac{180}{30}\) = 6
Molecular formula = \(C_6H_{12}O_6\)
(ii) The intermolecular bonding in ethanoic acid is hydrogen bonding while the butane molecules are only aggregated by weak van der Waals.
(a) (i) Polymerization is the combination of many molecules of the same kind to form a complex molecule Conc. H\(_2\)SO\(_4\) without gain or loss of material.
(b)(i) \(CH_3CH_2CH_2OH_{(aq)}\) \(CH_2CHCH_3\) + (\(H_2SO_4 + H_2O\)).
(ii) \(CH_3CH_2CH_2OH_{(aq)}\) + CHOOH --> \(CH_3CH_2CH_2OH_{(l)}\) + H\(_2\)O\(_{(l)}\)
(i) & (ii) are respectively dehydration and condensation type of reactions.
(c)(i) Carbon 40.O% = \(\frac{40}{12}\) = 3.33 moles ; Hydrogen 6.7% moles
\(\frac{6.7}{1} = 6.7\) moles
Oxygen 53.3% = \(\frac{53.3}{16}\) = 3.33 moles
Divide through by smaller number
i.e. C \(\frac{33}{3.33}\) : H \(\frac{6.7}{3.33}\) : O \(\frac{3.33}{3.33}\)
C = 1: H = 2.0: O = 1
Empirical formula = CH\(_2\)O(CH\(2\)O)x = 180
i.e. (30)x = 180
x = \(\frac{180}{30}\) = 6
Molecular formula = \(C_6H_{12}O_6\)
(ii) The intermolecular bonding in ethanoic acid is hydrogen bonding while the butane molecules are only aggregated by weak van der Waals.