(a) i) An organic compound X contains 40% carbon, 6.67% hydrogen, the rest being oxygen. If X has a relative molecular mass of 60, determine its
(i) empirical formula (ii) molecular formula. [H = 1 ; C = 12; O = 16]
(b) An alkanoic acid Y has a relative molecular mass of 74.
(i) State the functional group of Y
(ii) What t of reaction is involved when Y is converted to an alkanoate?
(iii) Determine the structural formula of Y.
(iv) Write an equation for the reaction between Y and sodium
(v) If X in (a) above boils at 118°C and belongs to the same homologous series as Y, state with reason, whether the boiling point of Y will be equal to, higher or lower than 118°C.
(c)(i) What is fermentation?
(ii) Write an equation for the fermentation of glucose.
(iii) What must be added to glucose solution to make it ferment?
(iv) Explain why a tightly corked grass bottle filled to the brim with fresh palm wine shatters on standing.
(i) empirical formula (ii) molecular formula. [H = 1 ; C = 12; O = 16]
(b) An alkanoic acid Y has a relative molecular mass of 74.
(i) State the functional group of Y
(ii) What t of reaction is involved when Y is converted to an alkanoate?
(iii) Determine the structural formula of Y.
(iv) Write an equation for the reaction between Y and sodium
(v) If X in (a) above boils at 118°C and belongs to the same homologous series as Y, state with reason, whether the boiling point of Y will be equal to, higher or lower than 118°C.
(c)(i) What is fermentation?
(ii) Write an equation for the fermentation of glucose.
(iii) What must be added to glucose solution to make it ferment?
(iv) Explain why a tightly corked grass bottle filled to the brim with fresh palm wine shatters on standing.
Take Free Practice Test On 2026 JAMB UTME, Post UTME, WAEC SSCE, GCE, NECO SSCE
Correct Answer: Option n
Explanation:
(a)(i) X contains 40% carbon, 6.67% hydrogen, the rest being oxygen. To determine the emperical formula of X
(i) Percentage oxygen = 100 – (40 + 6.67) = 53.33%
C : H O
40 6.67 53.33
12 1 16
3.33 6.67 3.33
1 2 1
Ratio
Empirical formula is CH\(_2\)O
(ii) Molecular formula of X if its relative molecular mass is 60. (CH\(_2\)O)\(_n\) = 60;
(12 + 2 + 16)\(_n\) = 60 ;
30n = 60 ; n = 2
Molecular formula is C\(_4\)H\(_4\)O\(_2\)
(b) Y is an alkanoic acid with relative molecular mass is 74.
(i) The functional group in Y is –COOH/carboxyl group.
(ii) Esterification reaction
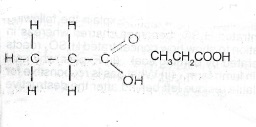
(iii) If Y is represented by C\(_n\)H\(_{2n+1}\)COOH; then 14n + 46 = 74, giving n = 2
(iv) 2C\(_2\)H\(_5\)COOH + 2Na –> 2C\(_2\)H\(_5\)COONa + H\(_2\).
(v) Compound Y in the same homologous series as X has a CH\(_3\)CH\(_2\)COOH relative molecular mass of 74. The boiling point of Y will be N higher. than 118° because in a homologous series boiling OH point increases with increase molar mass.
(c)(i) Fermentation is the breakdown of carbohydrates (sugars) to iorm ethanol (alcohol) and CO\(^2\) by the action of enzymes.
(iii) Yeast is adOed to glucose solution to make glucose ferment.
(iv) A tightly caked glass bottle filled to the brim with fresh palm wine shatters.on standing because CO\(^2\) is produced as the palm wine ferments. The gas causes pressure build-up which causes the bottle to shatter.
(a)(i) X contains 40% carbon, 6.67% hydrogen, the rest being oxygen. To determine the emperical formula of X
(i) Percentage oxygen = 100 – (40 + 6.67) = 53.33%
C : H O
40 6.67 53.33
12 1 16
3.33 6.67 3.33
1 2 1
Ratio
Empirical formula is CH\(_2\)O
(ii) Molecular formula of X if its relative molecular mass is 60. (CH\(_2\)O)\(_n\) = 60;
(12 + 2 + 16)\(_n\) = 60 ;
30n = 60 ; n = 2
Molecular formula is C\(_4\)H\(_4\)O\(_2\)
(b) Y is an alkanoic acid with relative molecular mass is 74.
(i) The functional group in Y is –COOH/carboxyl group.
(ii) Esterification reaction
(iii) If Y is represented by C\(_n\)H\(_{2n+1}\)COOH; then 14n + 46 = 74, giving n = 2
(iv) 2C\(_2\)H\(_5\)COOH + 2Na –> 2C\(_2\)H\(_5\)COONa + H\(_2\).
(v) Compound Y in the same homologous series as X has a CH\(_3\)CH\(_2\)COOH relative molecular mass of 74. The boiling point of Y will be N higher. than 118° because in a homologous series boiling OH point increases with increase molar mass.
(c)(i) Fermentation is the breakdown of carbohydrates (sugars) to iorm ethanol (alcohol) and CO\(^2\) by the action of enzymes.
(iii) Yeast is adOed to glucose solution to make glucose ferment.
(iv) A tightly caked glass bottle filled to the brim with fresh palm wine shatters.on standing because CO\(^2\) is produced as the palm wine ferments. The gas causes pressure build-up which causes the bottle to shatter.