A gaseous metallic chloride MCl consists of 20.22% of M by mass. The formula of the chloride is?
[ M = 27, Cl = 35.5]
[ M = 27, Cl = 35.5]
Take Free Practice Test On 2026 JAMB UTME, Post UTME, WAEC SSCE, GCE, NECO SSCE
Correct Answer: Option C
Explanation:
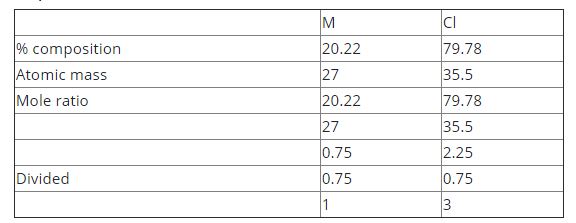
The formula of the Chloride = MCl\(_3\)
The formula of the Chloride = MCl\(_3\)