In the J-tube above, Y and X are on the same horizontal level and 30cm3 of air is trapped above Y when the atmospheric pressure is 75cm Hg, calculate the volume of air trapped above Y when 15cm Hg is now poured into the limb above X
Take Free Practice Test On 2026 JAMB UTME, Post UTME, WAEC SSCE, GCE, NECO SSCE
Correct Answer: Option B
Explanation:
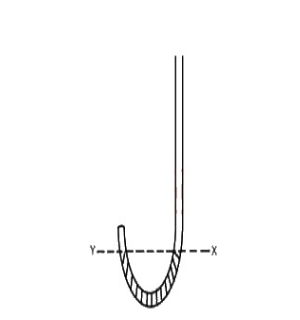

Since the temperature of the air is to remain Constant => P1V1 = P2V2
∴ 75 x 30 = (75 + 15) x V2
75 x 30 = 90 x V2
= 25cm2
Since the temperature of the air is to remain Constant => P1V1 = P2V2
∴ 75 x 30 = (75 + 15) x V2
75 x 30 = 90 x V2
| _90 |
| _90 |
= 25cm2